New Nanocomposite Enables Removal and Detection of Radioactive Iodine in Water
Researchers from Hefei Institutes of Physical Science, Chinese Academy of Sciences, have developed a silver-decorated, MOF-derived TiO₂₋ₓ nanocomposite enriched with oxygen vacancies, enabling both the removal and detection of trace radioactive iodide ions.
Their findings were published in Separation and Purification Technology.
Radioactive iodine is highly soluble in water, posing risks to public health and the environment. Existing materials for iodine treatment often face drawbacks such as easy oxidation and aggregation of silver nanoparticles, limited adsorption capacity, and dependence on bulky instruments for detection. These limitations hinder effective treatment of radioactive wastewater and rapid on-site monitoring.
In this study, the researchers used a titanium-based metal–organic framework, MIL-125, as a precursor to construct a multifunctional Ag₂O–Ag@TiO₂₋ₓ nanocomposite. Through a controlled pyrolysis and solution deposition strategy, the material was engineered with dual active sites of Ag⁰ and Ag₂O. The resulting structure integrates oxygen vacancies with an Ag/TiO₂₋ₓ Schottky junction, forming a synergistic system that enables iodide capture via a coupled photocatalytic oxidation and chemisorption mechanism.
Experimental results show that the nanocomposite delivers effective iodine adsorption and maintains stable removal performance even at low iodide concentrations. The treated water meets national surface water environmental standards. The material also exhibits iodoperoxidase-like activity, enabling a colorimetric sensing system based on AT/TMB/H₂O₂ for visual detection of iodide ions. The system shows good resistance to interference from common anions and performs reliably in complex water environments, including seawater and nuclear wastewater.
The material also shows good stability under irradiation. After exposure to γ-rays, it retains most of its adsorption performance, while the concentration of leached silver remains below national safety limits.
This study provides a practical approach for the removal and detection of radioactive iodine. It may support wastewater treatment in nuclear facilities and environmental remediation, particularly for long-lived isotopes such as iodine-129, which are commonly associated with nuclear power generation, spent fuel reprocessing, and nuclear accidents.
Article link:https://doi.org/10.1016/j.seppur.2026.137630
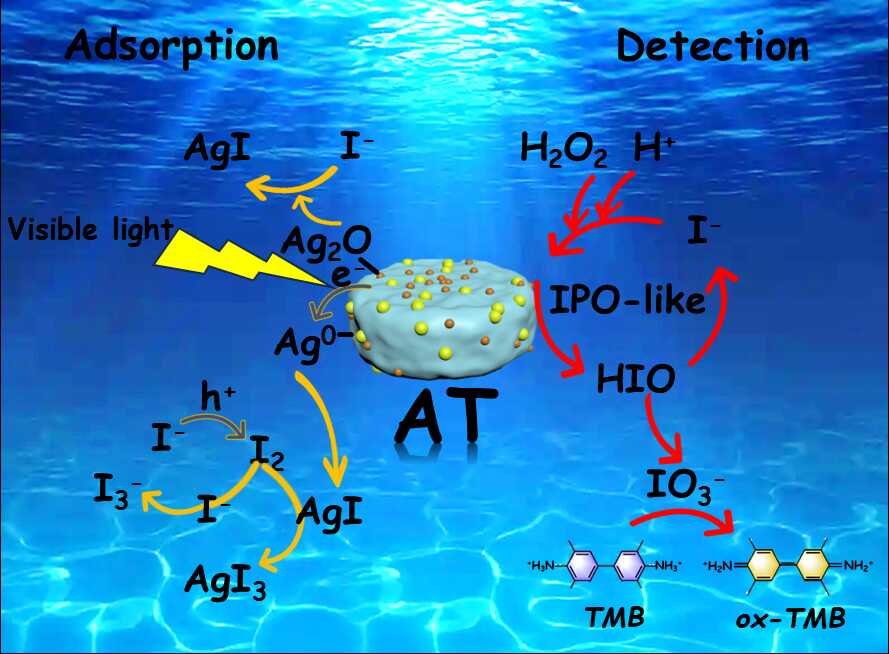
Figure 1. Adsorption and detection mechanism of radioactive iodide ions by AT nanocomposites.
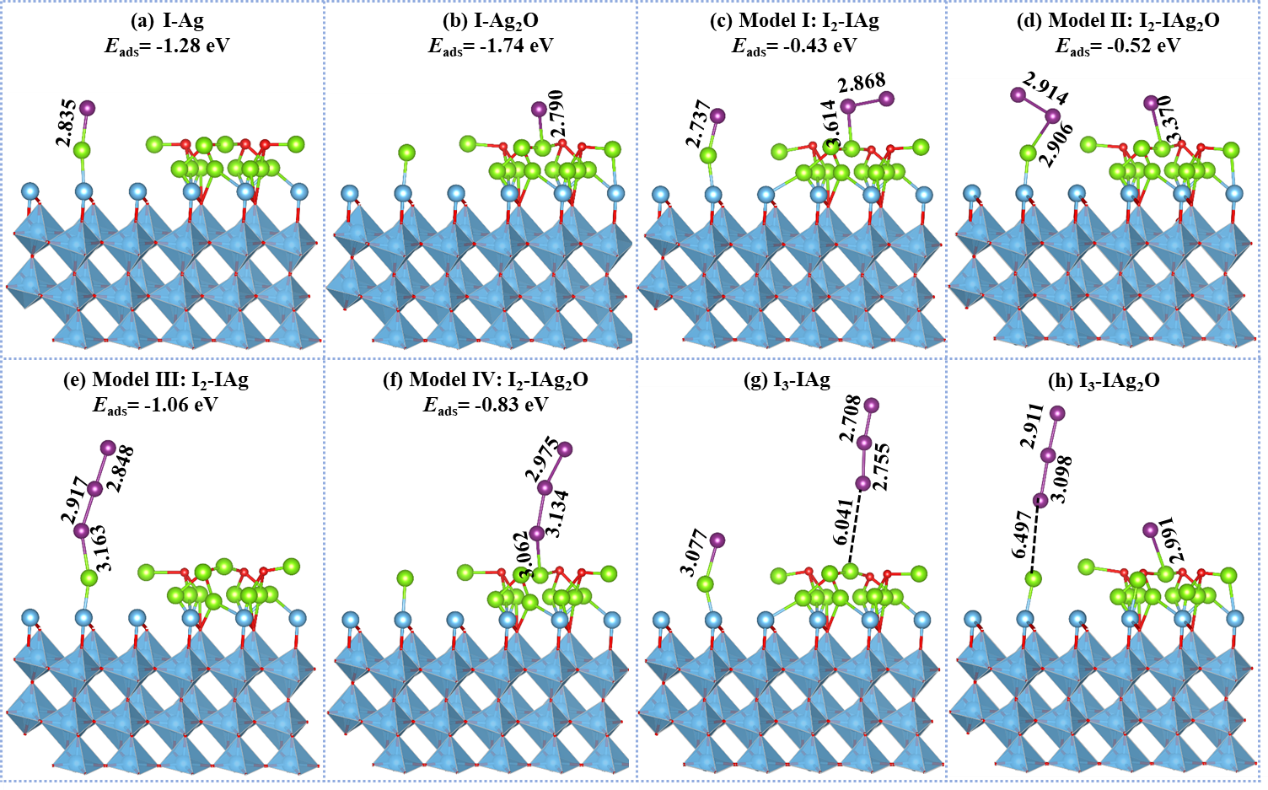
Figure 2. Optimized configurations of various iodine species adsorbed on adsorbent: (a-b) Adsorption of I- on AT nanocomposites; (c-f) Adsorption of I2 on I-AT nanocomposites; (g-h) Adsorption of I3- on I-AT nanocomposites.














