Novel Silica-based Adsorbent Helps Selective Separation of Strontium
Recently, Prof. HUANG Qunying’s team from Institute of Nuclear Energy Safety Technology(INEST) ,Hefei Institutes of Physical Science (HFIPS) of Chinese Academy of Sciences (CAS) developed a novel silica-based adsorbent for the highly selective separation of strontium from acidic medium, collaborating with Prof. NING Shunyan's team of South China University.
The results were published in Journal of Hazardous Materials.
Radioactive strontium (90Sr) is considered to be one of the most hazardous radionuclides due to its high biochemical toxicity. During the vitrification process of high level liquid waste (HLLW), the presence of 90Sr can cause instability of the vitrification substrate and thus induce radionuclides leaching. The removal of 90Sr can reduce the heat generation and shorten the cooling time of the vitrification substrate in the repository, which is helpful for further deep geological disposal of the radioactive waste.
To address the above issues, a novel porous CEPA@SBA-15-APTES adsorbent was prepared by the functionalized modification of mesoporous silica for the selective separation of Sr from acidic environment (i.e. 10-10-4 M HNO3 media), and the adsorption behavior of the adsorbent on Sr stable nuclide was investigated. The adsorbent possessed good acid resistance stability and adsorbed well on Sr stable nuclide. The adsorption equilibriums of Sr reached in 30 min for 10-6 M HNO3 and 5 min for 3M HNO3 media, respectively. The adsorption mechanisms of strontium were ion exchange in 10-6 M HNO3 solution and coordination in 3 M HNO3 solution.
This study developed a novel material for the efficient separation of strontium, which is a reference for the selective separation and recovery of radionuclides.
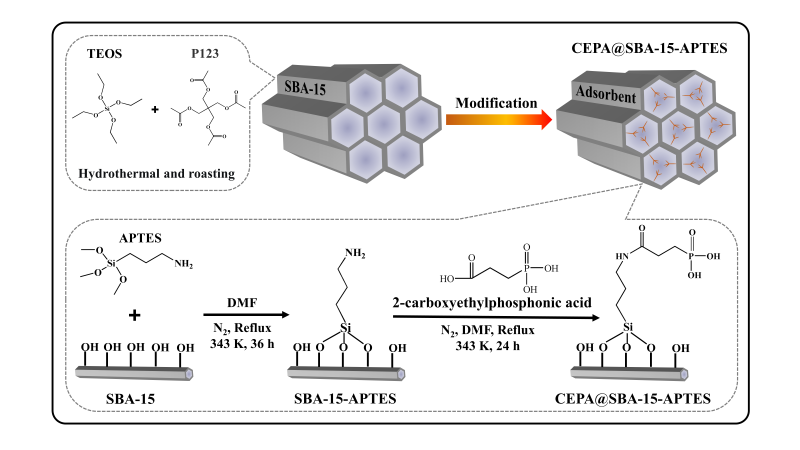
Figure 1 Schematic illustration of CEPA@SBA-15-APTES preparation process. (Image by ZHANG Shichang)
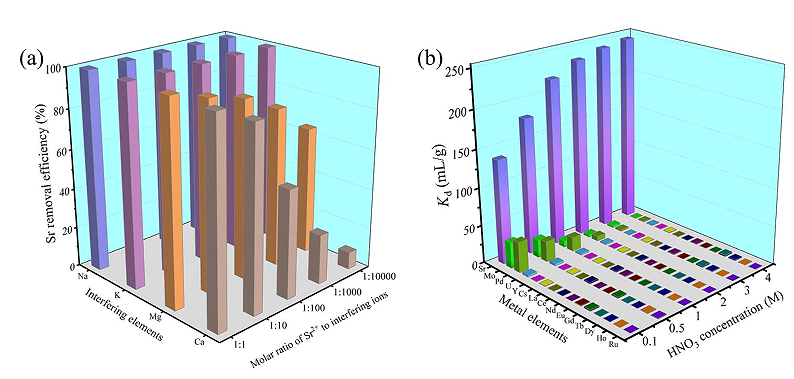
Figure 2 Adsorption selectivity: (a) Effect of four competing ions on Sr adsorption in 10-6 M HNO3 solution;
(b) Adsorption of different metal ions in the HNO3 solutions. (Image by ZHANG Shichang)














